Future Economic Trends: Forecasts for Generic Drug Markets
- Colin Hurd
- 27 February 2026
- 13 Comments
The global generic drugs market is on track to become one of the biggest forces in healthcare economics over the next decade. It’s not just about cheaper pills-it’s about making life-saving treatments accessible to millions who otherwise couldn’t afford them. As patents expire on blockbuster drugs like Humira, Enbrel, and now newer biologics like Dupixent and Skyrizi, a wave of affordable alternatives is flooding the market. This isn’t a passing trend. It’s a structural shift reshaping how the world pays for medicine.
Why Generic Drugs Are Growing So Fast
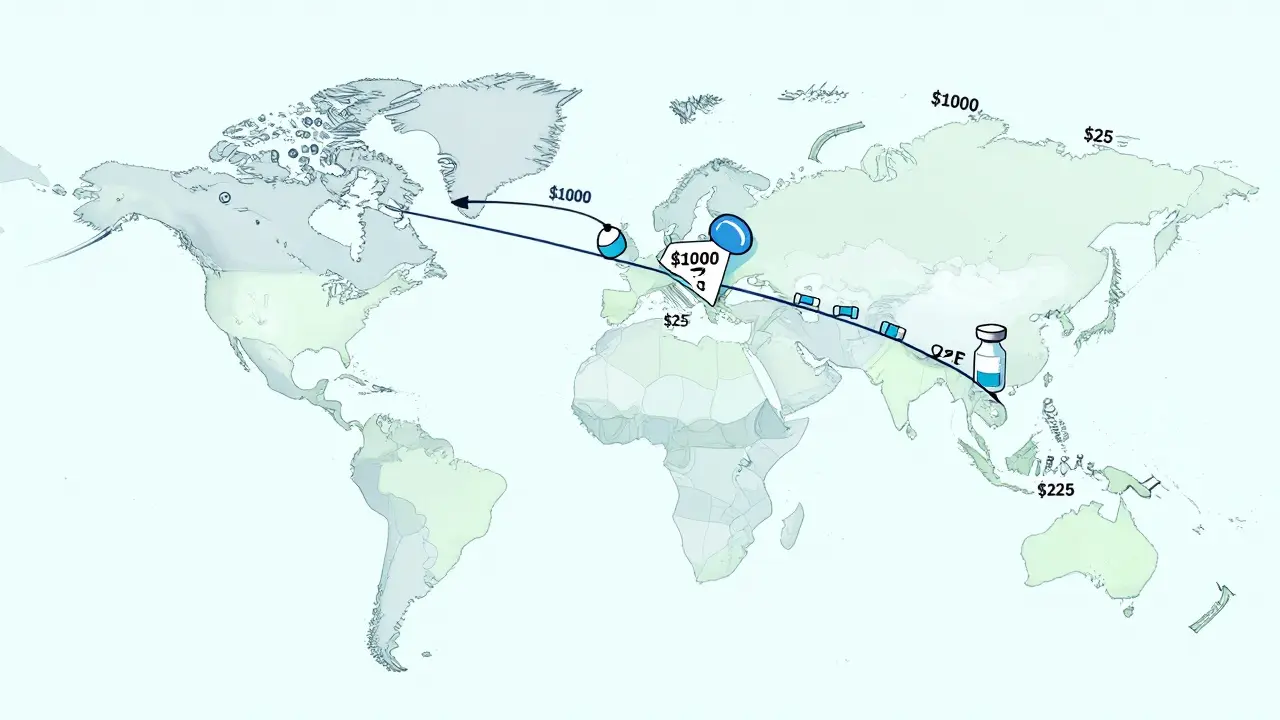
Between 2025 and 2030, drugs that made over $200 billion in annual sales will lose their patent protection. That’s not a typo. $200+ billion in revenue is about to open up for competitors. Companies that once had exclusive rights to sell these medications now face competition from manufacturers who can produce identical versions for a fraction of the cost. The result? Prices drop by 80% to 90% within months of a generic launch.
This isn’t happening by accident. Governments and insurers are pushing hard for generics because healthcare budgets are stretched thin. With aging populations in the U.S., Europe, and Japan, chronic conditions like diabetes, heart disease, and arthritis are becoming more common. Treating these conditions with branded drugs alone would bankrupt public health systems. Generic drugs are the only realistic way to keep treatment affordable.
Take diabetes, for example. The global number of people with diabetes is expected to hit over 700 million by 2030. Insulin, once priced at $1,000 per vial in the U.S., now has multiple generic versions available for under $25. That’s the power of competition. And it’s only starting.
The Rise of Biosimilars
While traditional generics copy simple chemical drugs, biosimilars are the next frontier. These are complex, biologically derived drugs-like monoclonal antibodies-that treat cancer, autoimmune diseases, and severe inflammation. They’re harder to make, harder to test, and cost more to develop than regular generics. But once they hit the market, they still slash prices by 30% to 70% compared to the original biologic.
Drugs like ustekinumab (Stelara) and vedolizumab (Entyvio) are losing patent protection between 2025 and 2027. That opens a $25 billion opportunity for biosimilar makers. Companies in India and China are already investing heavily in the technology to produce these drugs. Europe is leading the way with faster approval processes, and Japan is rushing to catch up with its own fast-track system. Early movers here will dominate the market for years.
Unlike simple generics, biosimilars aren’t exact copies. They’re highly similar, with minor differences that don’t affect safety or effectiveness. Regulatory agencies now accept this. The FDA and EMA have approved over 50 biosimilars since 2015, and the pace is accelerating.
Where the Growth Is Happening
The biggest growth isn’t in the U.S. or Europe-it’s in Asia. India alone supplies 20% of the world’s generic drugs and 60% of its vaccines. Indian manufacturers like Dr. Reddy’s, Sun Pharma, and Cipla have built massive, low-cost production networks. They don’t just make pills-they make them in bulk, with automated lines that cut waste and labor costs.
China is another powerhouse. Its government runs volume-based procurement tenders, where hospitals bid for the lowest price on bulk drug orders. This has forced global prices down. A generic version of a cancer drug that sold for $10,000 in the U.S. might be priced at $1,200 in China. Other countries now use China’s pricing as a benchmark.
But the fastest growth rate? Asia Pacific. Mordor Intelligence estimates the region will grow at 8.19% annually through 2030. Why? Rising incomes, expanding healthcare coverage, and growing awareness of generic quality. Countries like Indonesia, Vietnam, and Thailand are building local manufacturing. They’re not just importing generics-they’re making them.
Challenges in the Market
It’s not all smooth sailing. One major risk? Price erosion. In markets like the U.S., where insurers and pharmacy benefit managers (PBMs) negotiate aggressively, generic drug prices have been falling for years. Some pills now cost less than the packaging. That’s great for patients, but it squeezes profit margins for manufacturers. Many small players can’t survive.
Then there’s complexity. Older generics were easy to copy: a tablet with one active ingredient. Today’s generics include complex formulations-extended-release capsules, inhalers, injectables, and transdermal patches. These require advanced technology, strict quality control, and specialized equipment. Not every company can do it.
Supply chain issues also linger. The pandemic exposed how fragile global drug manufacturing is. Most active pharmaceutical ingredients (APIs) still come from China and India. Geopolitical tensions, export restrictions, or natural disasters can delay production. That’s why companies are now building dual-source manufacturing-keeping backup factories in different regions.
What’s Next? The Pipeline
The next big wave of generics will come from drugs used to treat obesity and metabolic disease. Semaglutide (Ozempic, Wegovy) and tirzepatide (Mounjaro, Zepbound) are among the most prescribed drugs in the U.S. today. They’re not yet off-patent, but they will be by 2030. When that happens, expect prices to plummet. A single monthly dose of Wegovy costs over $1,300. A generic version could drop below $100.
Also watch for GLP-1 agents-these are the same class of drugs as semaglutide. Early generic makers are already preparing. They’re investing in manufacturing lines that can handle the complex chemistry. Some are even partnering with tech companies to build digital tools that help patients stick to their treatment. Better adherence means more sales.
And don’t forget the role of automation. Robotic systems are now handling everything from pill counting to packaging. One Indian manufacturer reduced labor costs by 40% after installing automated lines. That’s not just efficiency-it’s survival.
Market Numbers: What the Experts Say
Estimates vary, but the consensus is clear: the market is growing.
- The global generic drugs market was valued between $488 billion and $491 billion in 2024.
- By 2030, most analysts project a market size between $640 billion and $700 billion.
- Some forecasters, like Towards Healthcare, predict $947 billion by 2034.
- The compound annual growth rate (CAGR) ranges from 4.2% to 8.5%, depending on assumptions.
- Biosimilars are growing faster-at 8.2% CAGR-outpacing traditional generics.
These numbers aren’t theoretical. They’re based on real patent expirations, real regulatory approvals, and real purchasing data from hospitals and pharmacies worldwide.
Who’s Winning?
The leaders aren’t the flashy names you see on TV. They’re the quiet manufacturers with factories in Hyderabad, Shanghai, and Guadalajara.
- India: Supplies 20% of global generics. Home to 500+ FDA-approved plants.
- China: Sets global price benchmarks through bulk procurement. Major exporter to Africa and Latin America.
- U.S. Players: Teva, Viatris, and Amneal dominate the domestic market but face pressure from overseas competition.
- Europe: Germany and the UK lead in adoption. Strong regulatory support and public trust in generics.
Smaller companies are getting squeezed. Only those with scale, regulatory expertise, and diversified supply chains will thrive.
What This Means for You
If you’re a patient, this is good news. You’ll pay less. You’ll have more options. You’ll get the same medicine, just cheaper.
If you’re a policymaker, this is a chance to stabilize healthcare spending. Generics reduce out-of-pocket costs for families and free up public funds for other needs-mental health services, preventive care, rural clinics.
If you’re an investor or manufacturer, the opportunity is huge-but so are the risks. The bar for entry is higher than ever. You need capital, technical skill, and regulatory savvy. But if you can navigate it, the returns are long-term and stable.
The future of medicine isn’t about inventing new drugs. It’s about making the existing ones accessible. Generic drugs are doing exactly that-and they’re just getting started.
Are generic drugs as effective as brand-name drugs?
Yes. In the U.S., the FDA requires generic drugs to have the same active ingredient, strength, dosage form, and route of administration as the brand-name version. They must also meet the same strict standards for quality, purity, and performance. Bioequivalence studies prove they work the same way in the body. Millions of patients use generics safely every day.
Why are generic drugs cheaper?
Brand-name companies spend billions on research, clinical trials, and marketing to get a drug approved. Generic manufacturers don’t have to repeat those costly studies. They only need to prove their product is equivalent. That cuts development costs by 80% or more. They also benefit from economies of scale-producing millions of pills at once-and lower marketing expenses.
Will generic drugs replace all brand-name drugs?
Not entirely. Some drugs, especially complex biologics and those with extended patents, will remain branded for years. But as patents expire, generics will take over the majority of prescriptions. In the U.S., generics already make up 90% of prescriptions but only 20% of spending. That ratio will keep growing.
How do biosimilars differ from generics?
Generics copy small-molecule drugs made from chemicals. Biosimilars copy large, complex biologic drugs made from living cells-like proteins or antibodies. Biosimilars are harder to produce, require more testing, and can have minor differences. But they’re not inferior. Regulatory agencies confirm they’re as safe and effective as the original.
What’s the biggest threat to the generic drug market?
Pricing pressure. In markets with aggressive bulk-buying systems-like China or the UK-prices can fall so low that manufacturers can’t profit. This risks supply shortages. Another threat is patent litigation. Some brand companies extend exclusivity through legal tactics, delaying generic entry. Regulatory delays and supply chain disruptions also pose risks, especially for complex drugs.

Comments
Charity Hanson
This is so cool to see! I grew up in Nigeria watching my mom struggle to afford insulin, and now seeing generics make it accessible? Tears. Not just for me-millions of moms like her are breathing easier. The real win isn’t the price drop-it’s the dignity it gives people. Keep pushing this forward, world.
Also, shoutout to Indian manufacturers. You’re not just making pills-you’re saving lives.
February 27, 2026 AT 14:32
Vikas Meshram
You people dont even understand how this works. The FDA approves generics but they are not identical. The excipients are different. And the bioavailability varies by up to 20%. That’s not a typo. 20%. And no one talks about it because Big Pharma pays the regulators. I’ve read the studies. The data is there. You just dont want to see it.
February 27, 2026 AT 19:02
Ben Estella
Let me get this straight-America spends trillions on healthcare while India makes our life-saving drugs for pennies? And we’re still whining about trade deficits? Wake up. This isn’t charity. This is competition. And we lost. The sooner we stop pretending our drug companies are heroes and start treating them like monopolies, the better off we’ll be.
February 27, 2026 AT 23:26
Jimmy Quilty
This is all a psyop. The WHO, FDA, and big pharma are in bed together to push generics so they can control the population with cheaper meds that have hidden additives. Why do you think they’re pushing biosimilars? They’re not safer-they’re *more* manipulable. And don’t get me started on how the supply chain is all routed through China. You think that’s coincidence? Think again.
March 1, 2026 AT 11:16
Miranda Anderson
I’ve been on a generic version of metformin for five years now. I’ve also taken the brand. Honestly? I can’t tell the difference. My blood sugar is stable. My doctor can’t tell the difference. My pharmacist says they’re bioequivalent. So why do we keep acting like generics are some kind of second-class option? It’s not about chemistry-it’s about perception. And that’s changing. Slowly. But it’s changing.
Also, the fact that we’re even having this conversation means we’re finally moving past the idea that expensive = better. That’s huge.
March 3, 2026 AT 05:23
Gigi Valdez
The structural shift described here is undeniable. The convergence of regulatory harmonization, manufacturing innovation, and fiscal necessity has created a rare alignment of incentives. What is particularly noteworthy is the transition from simple chemical generics to biosimilars, which introduces a new tier of complexity in quality assurance and supply chain integrity. The implications for global health equity are profound, though not without significant logistical challenges.
March 4, 2026 AT 22:40
Sneha Mahapatra
I just want to say… thank you to the workers in Hyderabad and Shanghai. People don’t think about them. The ones in the factories. The ones checking vials under microscopes at 3 a.m. The ones who never get a name on the label. This isn’t just economics. It’s quiet, invisible labor that’s holding the world together. 🙏
And yes, I cried reading about insulin dropping to $25. My cousin died because she couldn’t afford it. This matters.
March 6, 2026 AT 11:49
bill cook
I’m tired of this. You think generics are the answer? I’ve been on them. My anxiety got worse. My migraines got worse. I went back to the brand and I felt human again. So don’t tell me it’s all the same. You don’t know what it’s like to be a patient. You just know what the spreadsheet says.
March 6, 2026 AT 12:44
Byron Duvall
Okay, but who really owns the patents on these generics? It’s not India. It’s not China. It’s hedge funds. They buy the manufacturing rights, then flip them. The real money isn’t in making pills-it’s in buying the *right* to make them. And then they outsource to the lowest bidder. So yeah, pills are cheap-but the system? Still rigged.
March 6, 2026 AT 19:44
Brandon Vasquez
This is the quiet revolution. No hype. No viral tweets. Just factories running 24/7, regulators approving batches, pharmacists filling scripts. No one’s throwing a parade. But millions are alive because of it. That’s the kind of progress that doesn’t need applause.
March 7, 2026 AT 04:20
Full Scale Webmaster
Let’s be real. The entire system is a house of cards. The U.S. government pays $1,300 for Ozempic, then turns around and tells pharmacies to accept $100 generics. Who loses? The pharmacies. The manufacturers. The patients who get caught in the middle. And meanwhile, the PBMs? They’re still taking 20% off the top. This isn’t about affordability-it’s about redistribution of profit. And guess who’s not getting a cut? The people who need the medicine. I’ve seen the spreadsheets. This is a shell game with lives.
March 8, 2026 AT 22:13
Brandie Bradshaw
I appreciate the data, but let’s not romanticize this. Yes, generics save money. But the quality control inconsistencies? The batch failures? The lack of long-term post-market surveillance in low-regulation countries? We’re trading short-term cost savings for long-term public health risk. And when a batch of contaminated metformin causes kidney failure in 500 people across five countries? Who’s held accountable? The FDA? The Indian manufacturer? The U.S. insurer? No one. That’s not progress. That’s negligence dressed up as innovation.
March 8, 2026 AT 23:20
Eimear Gilroy
I’m from Ireland. We’ve been using generics for over a decade. Our system is simple: if it’s approved, it’s covered. No haggling. No middlemen. The result? Lower costs, better adherence, fewer hospitalizations. It works. Why can’t the U.S. just… copy this? It’s not rocket science.
March 9, 2026 AT 07:23